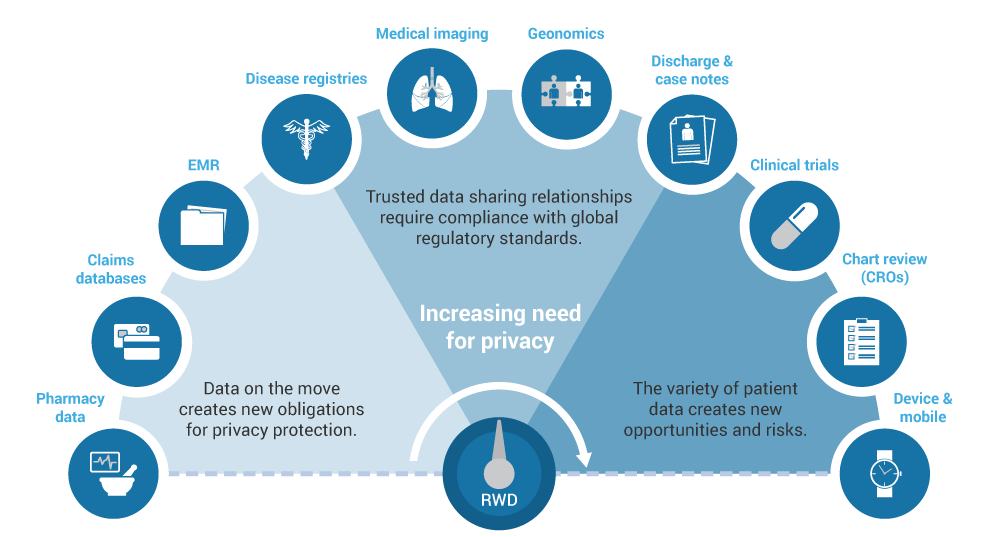
真實世界證據(jù)的增長需求
梅斯醫(yī)學(xué)的藥物安全和藥物警戒團(tuán)隊支持藥械的臨床試驗和上市后安全監(jiān)查,為我們客戶提供產(chǎn)品整個生命周期內(nèi)獨立和整體服務(wù)。
讓我們與眾不同的是我們的員工:高素質(zhì)的藥物安全科學(xué)家和熟練的醫(yī)療專業(yè)人員,他們在患者管理,特定的藥物警戒服務(wù)以及監(jiān)管法規(guī)方面,擁有深厚的專業(yè)知識和藥物安全管理豐富的經(jīng)驗。

我們可以協(xié)助您:
-
描繪自然病史和病程(例如,發(fā)病率,患病率,護(hù)理標(biāo)準(zhǔn))
-
通過描述疾病負(fù)擔(dān)來確定未滿足的臨床和人文需求
-
收集罕見疾病人群數(shù)據(jù)
-
量化真實世界產(chǎn)品特定和/或比較安全性,有效性,依從性和其他結(jié)局
-
評估特定的治療模式,量化相關(guān)的護(hù)理成本,并填充衛(wèi)生經(jīng)濟(jì)模型
深入的數(shù)據(jù)洞察力
我們專注于您的研究問題,并提供包含各種實際數(shù)據(jù)源和數(shù)據(jù)的最佳解決方案。我們由健康經(jīng)濟(jì)學(xué)家,流行病學(xué)家,生物統(tǒng)計學(xué)家和臨床醫(yī)生組成的綜合團(tuán)隊對可用的數(shù)據(jù)庫進(jìn)行嚴(yán)格評估,以確定那些能夠提供最佳信息以滿足您的研究需求的數(shù)據(jù)庫。我們對來自20多個國家(包括北美和歐洲)以及其他地區(qū)(如巴西,日本,韓國,中國,澳大利亞,臺灣)的數(shù)據(jù)來源有深入的了解和經(jīng)驗。我們了解并可以分析大型索賠數(shù)據(jù)庫和電子病歷以及這些和其他數(shù)據(jù)源的組合。
我們專注于您的研究問題,并提供包含各種實際數(shù)據(jù)源和數(shù)據(jù)的最佳解決方案。我們由健康經(jīng)濟(jì)學(xué)家,流行病學(xué)家,生物統(tǒng)計學(xué)家和臨床醫(yī)生組成的綜合團(tuán)隊對可用的數(shù)據(jù)庫進(jìn)行嚴(yán)格評估,以確定那些能夠提供最佳信息以滿足您的研究需求的數(shù)據(jù)庫。我們對來自20多個國家(包括北美和歐洲)以及其他地區(qū)(如巴西,日本,韓國,中國,澳大利亞,臺灣)的數(shù)據(jù)來源有深入的了解和經(jīng)驗。我們了解并可以分析大型索賠數(shù)據(jù)庫和電子病歷以及這些和其他數(shù)據(jù)源的組合。
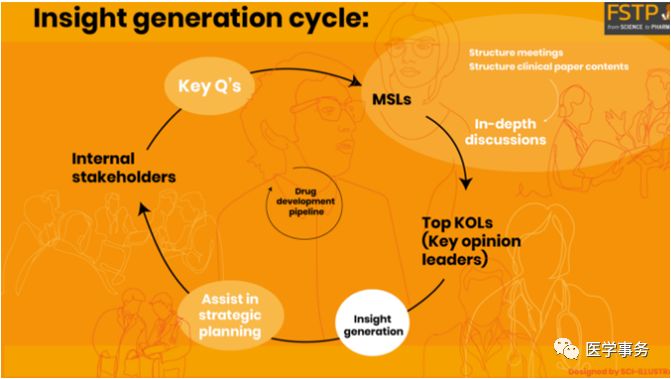
與專業(yè)機構(gòu)、協(xié)會、學(xué)會、Kols的深入合作
實現(xiàn)最佳市場準(zhǔn)入和有效商業(yè)化需要定制化的研究設(shè)計,深入的專業(yè)知識以及對全國Kols的關(guān)系網(wǎng)絡(luò)。我們對上市后環(huán)境的理解加上十?dāng)?shù)年臨床研究的經(jīng)驗,轉(zhuǎn)化為獨有的能力,可提供量身定制的有效研究設(shè)計,以滿足特定目標(biāo)和市場需求和產(chǎn)品的監(jiān)管要求。此外,我們的運營模式是根據(jù)醫(yī)療,臨床,項目管理,監(jiān)管,醫(yī)保和流行病學(xué)職能部門的專家意見量身定制的,以滿足項目特定目標(biāo)和利益相關(guān)者的期望。
卓越的研究設(shè)計和執(zhí)行
- 豐富的經(jīng)驗 - 專注于真實世界研究設(shè)計、運營、監(jiān)察和成果團(tuán)隊,15個重點臨床研究領(lǐng)域,200多個合作研究點,500多位長期合作研究者,7萬多主任醫(yī)師會員,200萬臨床醫(yī)師資源
- 專業(yè)的研究團(tuán)隊 - 在早期參與合作模式下工作時,在研究啟動和患者招募時間方面大大優(yōu)于行業(yè)基準(zhǔn)
- 治療領(lǐng)域?qū)<?- 具有廣泛適應(yīng)癥的能力,以及利用我們的臨床醫(yī)生和全國運營專業(yè)人員網(wǎng)絡(luò)的能力
- 通過技術(shù)和合作創(chuàng)新 - 充分利用人工智能和大數(shù)據(jù)技術(shù),以及真實世界研究的特點,大幅降低研究成本,提高操作的效率,確保高質(zhì)量的研究成果
上市后研究
專業(yè)團(tuán)隊為中國上市后研究助力
梅斯醫(yī)學(xué)真實世界研究BU在上市后臨床研究的設(shè)計和執(zhí)行方面具有廣泛的經(jīng)驗。我們的研究通過結(jié)構(gòu)化的研究設(shè)計和執(zhí)行產(chǎn)生有影響的醫(yī)學(xué)數(shù)據(jù),以及產(chǎn)品或特定患者群體的醫(yī)學(xué)證據(jù),以更好地指導(dǎo)臨床實踐,實現(xiàn)最市場準(zhǔn)入和最優(yōu)商業(yè),并滿足監(jiān)管當(dāng)局的要求。我們所有的研究都是按照GCP規(guī)范和相應(yīng)研究指南進(jìn)行的。
梅斯醫(yī)學(xué)開發(fā)針對上市后研究的精簡且經(jīng)濟(jì)有效的標(biāo)準(zhǔn)操作規(guī)程,最大限度地收集數(shù)據(jù)、監(jiān)控和質(zhì)量,同時最大限度地減少操作風(fēng)險。
梅斯醫(yī)學(xué)真實世界研究團(tuán)隊具有深厚的政策和專業(yè)知識,包括倫理和監(jiān)管要求、患者隱私立法、調(diào)查人員補助政策、學(xué)術(shù)和網(wǎng)站網(wǎng)絡(luò)、患者協(xié)會、國家衛(wèi)生數(shù)據(jù)庫和本地衛(wèi)生保健系統(tǒng)
卓越的研究設(shè)計與執(zhí)行
梅斯醫(yī)學(xué)真實世界研究BU在上市后臨床研究的設(shè)計和執(zhí)行方面具有廣泛的經(jīng)驗。我們的研究通過結(jié)構(gòu)化的研究設(shè)計和執(zhí)行產(chǎn)生有影響的醫(yī)學(xué)數(shù)據(jù),以及產(chǎn)品或特定患者群體的醫(yī)學(xué)證據(jù),以更好地指導(dǎo)臨床實踐,實現(xiàn)最市場準(zhǔn)入和最優(yōu)商業(yè),并滿足監(jiān)管當(dāng)局的要求。我們所有的研究都是按照GCP規(guī)范和相應(yīng)研究指南進(jìn)行的。
梅斯醫(yī)學(xué)開發(fā)針對上市后研究的精簡且經(jīng)濟(jì)有效的標(biāo)準(zhǔn)操作規(guī)程,最大限度地收集數(shù)據(jù)、監(jiān)控和質(zhì)量,同時最大限度地減少操作風(fēng)險。
梅斯醫(yī)學(xué)真實世界研究團(tuán)隊具有深厚的政策和專業(yè)知識,包括倫理和監(jiān)管要求、患者隱私立法、調(diào)查人員補助政策、學(xué)術(shù)和網(wǎng)站網(wǎng)絡(luò)、患者協(xié)會、國家衛(wèi)生數(shù)據(jù)庫和本地衛(wèi)生保健系統(tǒng)
藥品重點監(jiān)測
梅斯醫(yī)學(xué)真實世界研究BU在上市后臨床研究的設(shè)計和執(zhí)行方面具有廣泛的經(jīng)驗。我們的研究通過結(jié)構(gòu)化的研究設(shè)計和執(zhí)行產(chǎn)生有影響的醫(yī)學(xué)數(shù)據(jù),以及產(chǎn)品或特定患者群體的醫(yī)學(xué)證據(jù),以更好地指導(dǎo)臨床實踐,實現(xiàn)最市場準(zhǔn)入和最優(yōu)商業(yè),并滿足監(jiān)管當(dāng)局的要求。我們所有的研究都是按照GCP規(guī)范和相應(yīng)研究指南進(jìn)行的。
梅斯醫(yī)學(xué)開發(fā)針對上市后研究的精簡且經(jīng)濟(jì)有效的標(biāo)準(zhǔn)操作規(guī)程,最大限度地收集數(shù)據(jù)、監(jiān)控和質(zhì)量,同時最大限度地減少操作風(fēng)險。
梅斯醫(yī)學(xué)真實世界研究團(tuán)隊具有深厚的政策和專業(yè)知識,包括倫理和監(jiān)管要求、患者隱私立法、調(diào)查人員補助政策、學(xué)術(shù)和網(wǎng)站網(wǎng)絡(luò)、患者協(xié)會、國家衛(wèi)生數(shù)據(jù)庫和本地衛(wèi)生保健系統(tǒng)
Effective communication of evidence and information is essential to conveying the value of your products, both to internal and external stakeholders. With approximately 20 years of experience, our highly trained staff provide medical writing services for the peri- and post-approval phases of the product lifecycle, guaranteeing a consistency in quality and voice. Our writers prepare documents that are clear, concise, compelling, and scientifically accurate, while also ensuring they are fully compliant with regulations, industry best practices, and your corporate guidance.
- Site Identification
- KOLs networking
- PI Training
- Clinical Audit
- online SMO
Helping You Formulate a Market Access Strategy that Maximizes Access for Your Brand
With more expensive drugs winning regulatory approval and increased scrutiny on drug pricing, market access has never been more challenging and competitive. Our team offers a full spectrum of solutions and services to help you navigate the market access landscape and maximize access for your brand.
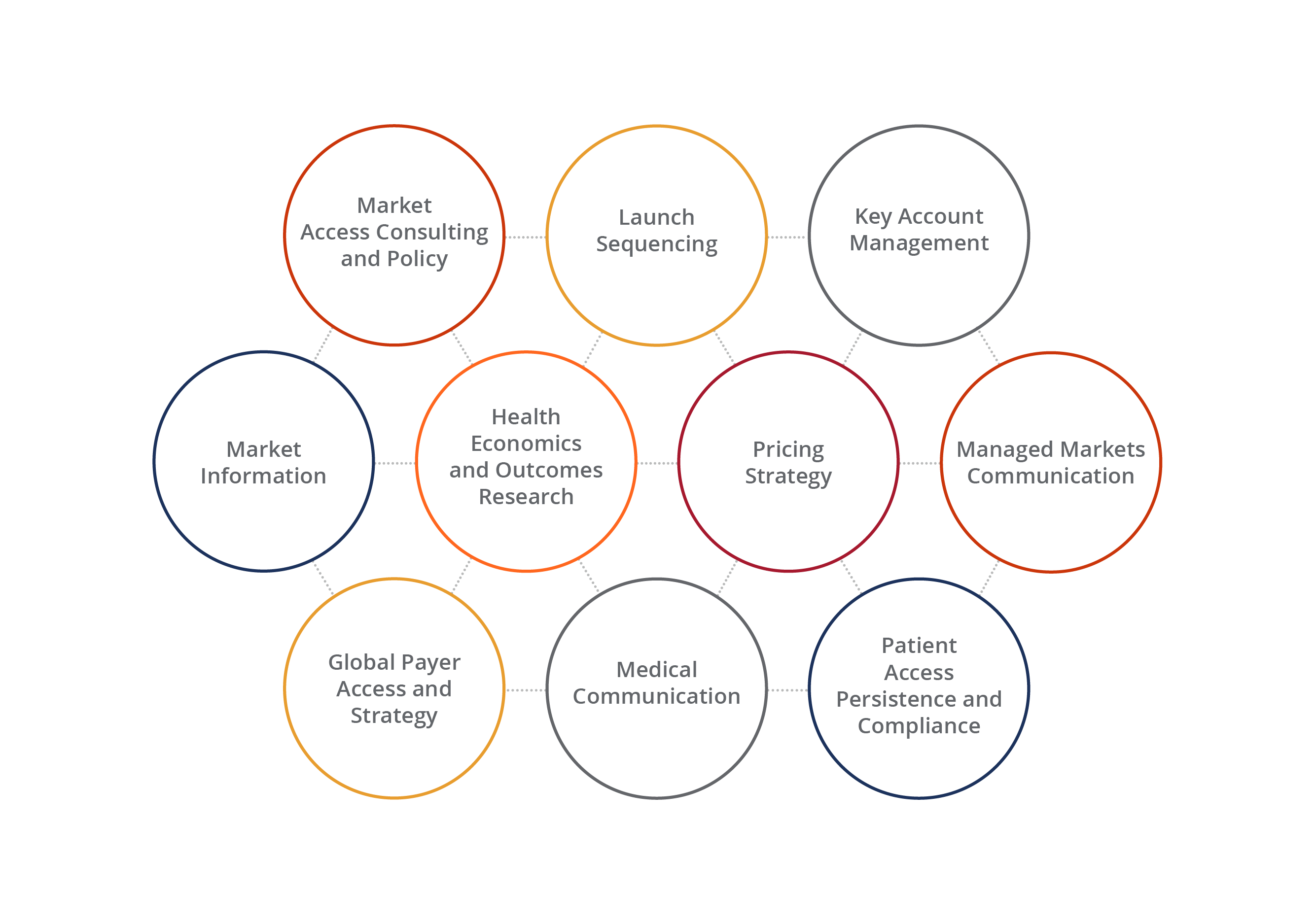
Optimizing Access to Markets Throughout Your Brand’s Life Cycle
Wherever you are in your brand’s life cycle, there is a team that can help you to achieve your access-related objectives. From research to strategy and tactical execution, we offer a comprehensive suite of services to give your brand and organization a competitive edge every step of the way.
A Complete Toolbox for Global Market Access
Whether it’s gathering market information or conducting cost analyses, we have the depth and resources to give our customers the insights they want and need. We bring these insights to life through our analytics, as well as our creative executions, generating captivating content that best communicates our customers’ strategy-driven messages.
?
Our Global Market Access Offering:
- Market assessment and analysis
- Comparative effectiveness research
- Pricing reimbursement and HEOR
- HCEI communications
- Patient assistance services
- Legislative and regulatory analysis
A Fit for All Shapes and Sizes
Whether you’re working for a company with a history of blockbuster launches or a start-up with a promising pipeline, we can help you develop and implement your market access strategy. Our multiple teams have collective experience across the healthcare continuum and can provide the support and services specific to your organization and brand, always tailoring them to help you overcome your brand-specific access challenges.
?
Close Connections on a Global Scale
With experienced teams in multiple countries, we understand the policies, trends and nuances in markets around the world.
BUILDING AND COMMUNICATING STRONG AND EFFECTIVE VALUE PROPOSITIONS
The complex, heterogeneous, and rapidly-changing payer environment is becoming increasingly difficult to navigate. Our expert teams of industry leaders, former payers, policy experts, and experienced strategists can help you navigate complex evidence requirements and anticipate and overcome payer barriers to help you achieve optimal price and access. Our team of communication specialists can help you develop compelling arguments presented in an accurate, succinct, and engaging story format to shape effective payer positioning and position evidence to overcome payer objections.
WE CAN HELP YOU
Gather in-depth information on the HTA, pricing, and reimbursement drivers and limiters, as well as landscape reviews, analog and market assessments
- Develop comprehensive global pricing and market access strategies and recommendations
- Develop evidence generation strategies to support optimal value positioning, HTA assessment, and pricing
- Support in-licensing decisions with rapid turnaround insights into global pricing and reimbursement potential
- Provide cross-functional alignment on pricing, access and evidence strategies through market access workshops
- Craft value stories that articulate a logical flow of arguments, supported by available evidence
- Test, refine, and validate value messages with relevant stakeholders
- Present payer-relevant evidence through global value dossiers, slide decks, objection handlers, and training
- Develop country-specific pricing and reimbursement or formulary submissions, including Academy of Managed Care Pharmacy (AMCP) dossiers for U.S. payers and HTA submissions for various markets
- Convey messages and evidence in a dynamic, user-friendly way electronically through?iValue Suite
- Explore key market access hypotheses through international payer advisory boards
WE OFFER YOU
- A network of over 2,000 payer experts and advisors worldwide
- A Pricing and Reimbursement Policy Council (PRPC) comprised of a diverse group of industry experts, and providing insights into the latest issues, trends, and changes affecting market access
- Expertise and experience in over 45 countries and across all major therapeutic areas
- In-house expertise and no outsourcing, with ~35,000 payer interviews and all value story and dossier writing done internally
- Rigorous quality standards applied to all content development
ADDRESSING THE LATEST TRENDS IN MARKET ACCESS
- Advanced Therapy Medicinal Products
- Early Access
- Expanded Access
- Conditional Marketing (e.g,, for Orphan Drugs)
- Medical Devices
- Price Referencing
- Surrogate Endpoint Use
- Transparency in Drug Pricing
Effective communication of evidence and information is essential to conveying the value of your products, both to internal and external stakeholders. With approximately 20 years of experience, our highly trained staff provide medical writing services for the peri- and post-approval phases of the product lifecycle, guaranteeing a consistency in quality and voice. Our writers prepare documents that are clear, concise, compelling, and scientifically accurate, while also ensuring they are fully compliant with regulations, industry best practices, and your corporate guidance.
- Real-world data collection
- patient or physician surveys
- Hybrid database studies
- Product Registries
- Disease Registries
- Electronic clinical outcome assessment eCOA (ePRO/eClniRO/eObsRO)